FDA Approves New Version of PrEP—Just Two Shots A Year
The FDA has approved lenacapavir as a form of pre-exposure prophylaxis (PrEP), offering a new option for HIV prevention requiring only two shots per year.
The United States Preventative Service Task Force (USPSTF) recently released updated recommendations on Pre-Exposure Prophylaxis for preventing the sexual transmission of HIV. The recommendations add additional medications, including a long-acting injectable, to the possible regimens and gives these strategies an A grade which should help eligible individuals get insurance coverage for PrEP.
PrEP was introduced in 2012 when the FDA approved Truvada, a daily pill for people who are HIV-negative but at higher risk of infection. If taken as directed, Truvada can lower the risk of contracting HIV by up to 99%. In 2019, the USPSTF gave Truvada as PrEP an A grade. Since that time new medications for PrEP have been approved including generic versions of Truvada, Descovy (another oral medication), and cabotegravir (an injectable drug given every two months that is marketed as Apretude). The updated recommendations released last week add all of these options to the A grade.
The Task Force makes recommendations based on weighing the benefits and harms of a medical intervention. While it does not consider insurance coverage, the grades have implications because the Affordable Care Act says that private insurers must cover preventative services that receive an A or B grade with no co-pay or deductible. (That provision of the ACA is being challenged in court by a group of businesses and individuals who say that they should not be forced to pay for preventative services, like PrEP, that violate their religious beliefs. A lower court in Texas has ruled in their favor and the case is now being appealed.)
The Task Force’s recommendations also include a discussion of who should have access to PrEP. They suggest that providers ask all patients about their sexual and injection drug use history and offer PrEP to sexually active adults and adolescents who have engaged in anal or vaginal sex in the past 6 months, and:
The recommendations also suggest that clinicians offer PrEP to anyone who injects drugs or has a partner with HIV who injects drugs.
These recommendations vary slightly from CDC guidelines which suggest providers discuss PrEP with all sexually active adults and adolescents and prescribe to anyone who asks for it.
Uptake of PrEP since it was introduced has been disappointing to many public health experts who see this prevention strategy as key to ending the epidemic. The CDC estimates that 1.2 million people are eligible for PrEP but only 30% of them are actually using it. Moreover, there are significant disparities in who is accessing this prevention method. While 78% of white people who are eligible for PrEP are taking it, only 11% of eligible Black people, 12% of eligible women, and 21% of eligible Latinas/os are using PrEP.
As the Task Force explained in an accompanying article for the Journal of the American Medical Association (JAMA), “PrEP is the ultimate implementation science challenge. At more than 90 percent efficacy when used properly, the only barrier to drastically reducing HIV incidence in the U.S. is addressing the systemic low coverage.”

The FDA has approved lenacapavir as a form of pre-exposure prophylaxis (PrEP), offering a new option for HIV prevention requiring only two shots per year.

A new study found that opt-out screenings for all patients in emergency departments caught numerous cases of syphilis and HIV that would have gone undetected under other screening protocols.
HIV stands for Human Immunodeficiency Virus. Learn the basics about HIV prevention, testing, and treatment.

PrEP stands for pre-exposure prophylaxis. The word prophylaxis means to prevent or control the spread of an infection or disease. The goal of PrEP is to prevent HIV infection from taking hold if you are exposed to the virus.

Life continues after becoming HIV positive. Thanks to new treatments, many people with HIV are living long, healthy lives.

Two studies published in this month’s JAMA Internal Medicine offer encouraging findings about the use of Doxy PEP.
Remarkable results in a Phase III PrEP trial with women and adolescent girls in Africa—two injections per year effectively prevented all HIV infection.
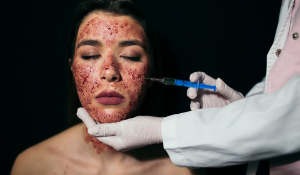
A joint investigation by the CDC and the New Mexico Health Department has confirmed that an unlicensed medi-spa is responsible for at least three cases of HIV.
ASHA believes that all people have the right to the information and services that will help them to have optimum sexual health. We envision a time when stigma is no longer associated with sexual health and our nation is united in its belief that sexuality is a normal, healthy, and positive aspect of human life.
ABOUT
GET INVOLVED
ASHA WEBSITES
GET HELP
© 2026 American Sexual Health Association
We need to know if we can keep you company during this visit. We are useful for making this site work.
We use cookies to enhance your browsing experience. You can choose which cookies you want to accept.
Necessary cookies help make a website usable by enabling basic functions like page navigation and access to secure areas. The website cannot function properly without these cookies.
| Cookie | Provider | Purpose | Expiry |
|---|---|---|---|
digiconsent | This website | Stores your cookie consent preferences. | 1 year |
wordpress_logged_in_* | WordPress | Identifies logged-in users and their authentication details. | 14 days / Session |
wordpress_sec_* | WordPress | Stores authentication details for secure areas. | 14 days / Session |
wp-settings-* | WordPress | Stores user interface customization preferences. | 1 year |
wp-settings-time-* | WordPress | Stores the time when wp-settings cookie was set. | 1 year |
Analytics cookies help us understand how visitors interact with our website by collecting and reporting information anonymously. This helps us improve our website.
| Cookie | Provider | Purpose | Expiry |
|---|---|---|---|
_ga | Registers a unique ID to generate statistical data on website usage. | 2 years | |
_ga_* | Used by Google Analytics to store and count pageviews. | 2 years |
Marketing cookies are used to track visitors across websites. The intention is to display ads that are relevant and engaging for the individual user.
Functional cookies enable the website to provide enhanced functionality and personalization. They may be set by us or by third party providers.