
HPV Screening with Self Collection Endorsed by Three Organizations
Three organizations—ACS, ACOG, and HRSA—have each recently endorsed new guidelines for cervical cancer screening. All of them discuss screening with self collected samples.
In the U.S., HPV infections are estimated to cause about 37,300 cases of cancer. HPV vaccination can prevent over 90% of these cancers from ever developing.
The HPV vaccine available in the U.S. has been approved for the prevention of cervical, anal, vulvar and vaginal cancers and pre-cancers. The vaccine protects against seven types of “high-risk” HPV that are associated with cancer. It also protects against two “low-risk” types associated with genital warts.
Rates of infection with strains of HPV covered by the vaccines have dropped significantly in the U.S. and around the world since the vaccine was introduced.
HPV vaccination is recommended for girls and boys ages 11-12. Why so young? For one, the vaccine produces a stronger immune response when taken during the preteen years, and fewer shots are required. Also, the vaccine is designed to prevent infection, so vaccination is recommended at a young age—before kids are exposed to the virus.
While the recommended age for HPV vaccine is 11-12, vaccination can start as early as age 9. Youth between ages 9 and 14 only need two doses of the HPV vaccine. The schedule calls for the second dose of the vaccine to be given 6-12 months following the first.
For older teens and adults who start the series later, starting at age 15, three doses are still required. In this case, the second shot should be given one to two months after the first, and the third shot should be given six months after the first. The goal is to get all three shots within six months.
In late 2018, the FDA approved use of the HPV vaccine in all people ages 27-45, expanding the previous indication that covered from ages 9-26. Ideally, people should complete all doses of the vaccine before they become sexually active. However, adults over 27 now have the option of receiving the vaccine and should talk to their health care provider to make the decision about vaccination.
The HPV vaccine is safe. Over 135 million doses of the HPV vaccine have been distributed in the U.S. and over 270 million have been distributed worldwide since the vaccine was licensed, and data has shown it to be safe and well tolerated.
The most common side effect of the vaccine is pain, redness, and swelling around where the shot was given. Other mild reactions reported include fever, headache, fatigue, nausea and vomiting. Some people have experienced fainting as well.
As with any vaccine or medication, there is always a possible of a serious problem, such allergic reaction. However, these reactions are rare. The HPV vaccine also continues to be monitored for any safety concerns.
The vast majority of health insurance plans report including most or all of the ACIP recommended vaccines in their benefits for children adolescents and adults.
For those that qualify, HPV vaccines are also available through the federal Vaccines for Children (VFC) program. The VFC program provides free vaccinations for children aged 18 and under who meet at least one of the following criteria: 1) Medicaid eligible; 2) uninsured; 3) underinsured; or 4) Native American or Alaska Native.
No. While some parents have expressed a concern that vaccinating their children against HPV will lead to increased sexual behavior, several studies have shown this not to be the case.
Yes. HPV vaccines will not eliminate all HPV, cervical cancer, or other HPV-related cancer. The vaccines prevent the HPV types that cause 70% of cervical cancer cases, but there are other types of HPV (not covered in the vaccine) that could cause disease.
Cisgender women and transgender men who still have a cervix should still screen regularly with Pap and/or HPV tests so any problems can be diagnosed and treated early.

Three organizations—ACS, ACOG, and HRSA—have each recently endorsed new guidelines for cervical cancer screening. All of them discuss screening with self collected samples.
Research suggests that only one dose of the HPV vaccine may be effective enough to prevent HPV-related disease, including cancer.

Australia has been a leader in cervical cancer prevention for decades and is now close to eliminating the disease.

The American Cancer Society (ACS) released new recommendations for cervical cancer screening that focus on HPV testing and approve the use of self-collected samples. The recommendations also clarify the age at which screening should start and stop.
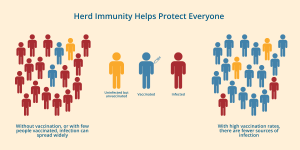
We’ve known for years that the HPV vaccine works. Now new research shows that widespread vaccination even protects those who haven’t gotten the shot. This study proves that it is possible to reach herd immunity for HPV.

After a diagnosis, it’s normal to have questions about HPV and relationships: What does this mean for our health? Did someone cheat? Should we stop having sex?

The FDA just approved the Teal Wand, a self-collection device for HPV testing that does not require a speculum exam or even a trip to the doctor’s office. People can collect their own sample at home and send it to a lab for analysis.

The results of large-scale study of HPV suggest that one shot of the vaccine may be enough to protect young people from HPV and related issues like cervical cancer.
ASHA believes that all people have the right to the information and services that will help them to have optimum sexual health. We envision a time when stigma is no longer associated with sexual health and our nation is united in its belief that sexuality is a normal, healthy, and positive aspect of human life.
ABOUT
GET INVOLVED
ASHA WEBSITES
GET HELP
© 2026 American Sexual Health Association
We need to know if we can keep you company during this visit. We are useful for making this site work.
We use cookies to enhance your browsing experience. You can choose which cookies you want to accept.
Necessary cookies help make a website usable by enabling basic functions like page navigation and access to secure areas. The website cannot function properly without these cookies.
| Cookie | Provider | Purpose | Expiry |
|---|---|---|---|
digiconsent | This website | Stores your cookie consent preferences. | 1 year |
wordpress_logged_in_* | WordPress | Identifies logged-in users and their authentication details. | 14 days / Session |
wordpress_sec_* | WordPress | Stores authentication details for secure areas. | 14 days / Session |
wp-settings-* | WordPress | Stores user interface customization preferences. | 1 year |
wp-settings-time-* | WordPress | Stores the time when wp-settings cookie was set. | 1 year |
Analytics cookies help us understand how visitors interact with our website by collecting and reporting information anonymously. This helps us improve our website.
| Cookie | Provider | Purpose | Expiry |
|---|---|---|---|
_ga | Registers a unique ID to generate statistical data on website usage. | 2 years | |
_ga_* | Used by Google Analytics to store and count pageviews. | 2 years |
Marketing cookies are used to track visitors across websites. The intention is to display ads that are relevant and engaging for the individual user.
Functional cookies enable the website to provide enhanced functionality and personalization. They may be set by us or by third party providers.