
Mpox Passed on Without Symptoms? New Research Shows It’s Possible
Is mpox passed on without symptoms? In a new study of more than 8,000 men, about 1% of the men tested positive despite never having had symptoms.

According to new data from the Centers for Disease Control and Prevention (CDC), only one in three adults in the U.S. infected with hepatitis C have been cured despite the introduction of direct-acting antivirals almost 10 years ago.
Hepatitis is an inflammation of the liver. It can be caused by a group of viruses—hepatitis A, B, C, D and E. These viruses can damage liver cells and cause scar tissue. Symptoms can range from mild (fatigue) to more severe (mental confusion). In many cases hepatitis is not a serious threat to health and can resolve on its own. For some people, though, the disease can become long-lasting and may lead to liver failure and death.
Hepatitis C virus (HCV) is primarily transmitted by direct contact with blood. It can be transmitted through sharing needles or other injecting equipment during intravenous drug use. Having an STI or HIV, having sex with multiple partners, or engaging in rough sex may increase the likelihood that a person will contract HCV.
Until about 10 years ago, treatment for HCV involved versions of interferon. These treatments often came with unpleasant side effects. They were also not considered a cure, as a significant percentage of patients relapsed.
In 2013, Gilead introduced Solvadi, a 12-week course of treatment that cured HCV in up to 96% of patients. While this was a major breakthrough, people were outraged by the $84,000 price tag which prompted a Congressional investigation. Today, there are a number of similar medications that are nearly 100% effective in curing HCV. This includes lower-priced generic versions, but even these can cost over $20,000 without insurance.
A new study published in Morbidity and Mortality Weekly Report analyzed data from patients tested for hepatitis C. Over 1.7 million patients who had received a positive HCV antibody test were included. Of those that had an initial infection and were eligible for treatment, 34% were cured (with medication) or cleared (spontaneously). Among this group, however, 7% were later found to have a persistent infection or reinfection.
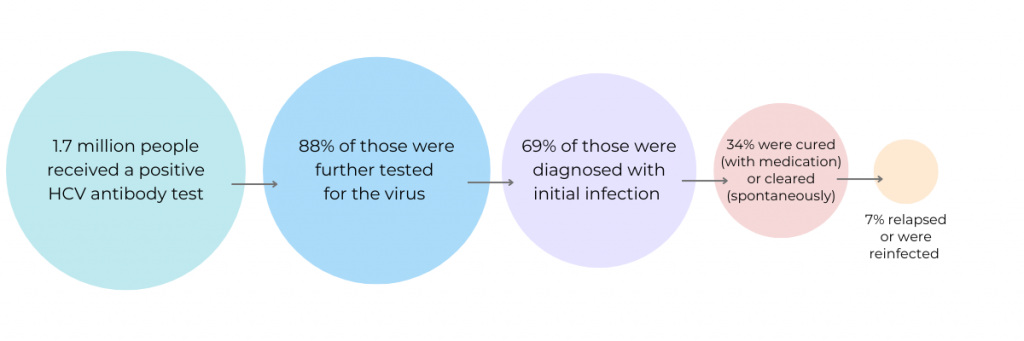
The researchers then looked at data by age, sex, and type of insurance. Not surprisingly, patients with other payor, unspecified payor/insurance, or Medicaid had lower viral clearance (23%, 33%, and 31%, respectively) than those with Medicare and commercial insurance (40% and 45%, respectively).
At a White House briefing on the topic, lead research Carolyn Wester called the cure rates “jarringly low.” She suggested the high price of treatment and numerous restrictions put in place by insurance companies were partially to blame. Some payers limit which patients are eligible for treatment. They may also require burdensome pre-authorization before treatment can begin. Insurers may even limit the types of providers who can prescribe treatment. All of these restrictions can delay or even prohibit access to this life-saving medication.
Other experts pointed point to the cumbersome testing process. The two-step process requires patients to get their results and then come for a second confirmatory test. This can lead to delays in treatment for some people or no treatment for those who don’t follow up. (The CDC recommends one-time hepatitis C testing of all adults 18 years and older. It also suggests that all pregnant women get tested during every pregnancy.)
Francis Collins, who is leading the White House National Hepatitis C Elimination Program, addressed this at the briefing. He argued that the U.S. needs rapid point-of-care tests like those already available in Europe and Australia. Collins suggested that initiating a rapid testing program could save the $44 billion over the next 20 years. It’s rare to have both an opportunity to save lives and save money, but that’s what we have here. You can’t know this and just walk away, he said.
The Viral Hepatitis National Strategic Plan calls for more than 80% of people with hepatitis C to achieve viral clearance by 2030. This study shows that we are still far from reaching that goal.

Is mpox passed on without symptoms? In a new study of more than 8,000 men, about 1% of the men tested positive despite never having had symptoms.

New data shows that cases of bacterial STIs have dropped dramatically in San Francisco. Public health officials in the city believe doxy PEP has helped drive these declines.

A new report shows that fewer women are getting prenatal care. That means too many patients are missing the chance to prevent congenital syphilis.

Condoms. All sorts of shapes, sizes, colors (even glow-in-the-dark). Ribbed or plain. Lubed or not. Latex or plastic. ASHA’s Fred Wyand shares his thoughts on this reliable way to prevent STIs and pregnancy.
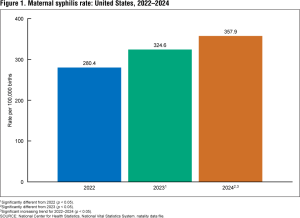
Syphilis rates in pregnant women went up 222% percent between 2016 and 2022 and another 28% between 2022 and 2024. Too many pregnant women are not being screened.

Three organizations—ACS, ACOG, and HRSA—have each recently endorsed new guidelines for cervical cancer screening. All of them discuss screening with self collected samples.
ASHA believes that all people have the right to the information and services that will help them to have optimum sexual health. We envision a time when stigma is no longer associated with sexual health and our nation is united in its belief that sexuality is a normal, healthy, and positive aspect of human life.
ABOUT
GET INVOLVED
ASHA WEBSITES
GET HELP
© 2026 American Sexual Health Association
We need to know if we can keep you company during this visit. We are useful for making this site work.
We use cookies to enhance your browsing experience. You can choose which cookies you want to accept.
Necessary cookies help make a website usable by enabling basic functions like page navigation and access to secure areas. The website cannot function properly without these cookies.
| Cookie | Provider | Purpose | Expiry |
|---|---|---|---|
digiconsent | This website | Stores your cookie consent preferences. | 1 year |
wordpress_logged_in_* | WordPress | Identifies logged-in users and their authentication details. | 14 days / Session |
wordpress_sec_* | WordPress | Stores authentication details for secure areas. | 14 days / Session |
wp-settings-* | WordPress | Stores user interface customization preferences. | 1 year |
wp-settings-time-* | WordPress | Stores the time when wp-settings cookie was set. | 1 year |
Analytics cookies help us understand how visitors interact with our website by collecting and reporting information anonymously. This helps us improve our website.
| Cookie | Provider | Purpose | Expiry |
|---|---|---|---|
_ga | Registers a unique ID to generate statistical data on website usage. | 2 years | |
_ga_* | Used by Google Analytics to store and count pageviews. | 2 years |
Marketing cookies are used to track visitors across websites. The intention is to display ads that are relevant and engaging for the individual user.
Functional cookies enable the website to provide enhanced functionality and personalization. They may be set by us or by third party providers.